Chemical Buffers in Hydroponics : What is the Best, Cheapest Buffer
From the very large library of chemical buffers available to the modern chemist, only a handful are suitable for their use in hydroponics and – even then – most of these are actually not practical in the sense that they are extremely expensive for the home grower. Certain organic buffers like MES offer extremely good results although they are hard to buy and very expensive, a reason why they are not used widely in hydroponic culture (this buffer is used mainly on hydroponic research where precision – and not cost – is the main determinant factor).
The buffers we are left with are then very simple organic and inorganic substances that have low phytotoxicity and some compatibility with the other ions present in our hydroponic setup. From these ions phosphate species, citrate species and carbonates are the most important ones we can use within our hydroponics setup. However we are limited by the actual concentration values of each we can use and for this reason we cannot have unlimited buffering capacity from these sources.
Which one is best ? We can actually carry out simulations to show us the pH vs acid-base addition for different hydroponic solution constitutions using mathematical equations. Running these simulations requires the solution of highly complex systems of equations which contain all the information relative to the chemical equilibrium of all the different existing ionic species. The below shown simulations were carried out using the Mathematica computer program (all solutions are assumed to be adjusted to an initial pH of 5.8 with a strong acid or base).
– –
–
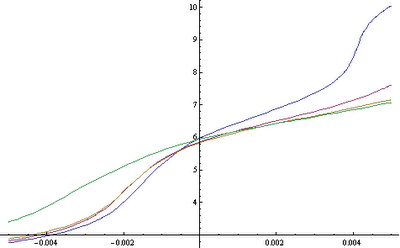
The blue curve represents the behavior of a poorly buffered hydroponic solution with only about 0.002M phosphate concentration (about 50 ppm of P). The red and yellow curves represent two solutions with increasing levels of carbonate showing us that if you are battling pH increases, having more carbonate will definitely help you deal with this. However it is also clear that carbonate concentrations at pH 5.8 are restricted to around 100 ppm since values above this are bound to cause toxicity due to the very large presence of the hydrogen carbonate ion. The green curve represents an increase in the amount of phosphate from 0.002 to 0.004M (about 100 ppm) with carbonate, showing us that phosphates are not good at buffering increases towards the upper side but they do increase buffering towards acid territory. Overall I also noticed that citrate concentration increases to the maximum threshold allowed by calcium citrate solubility did not afford a very good buffering effect with only a mild effect that prevented shifts towards the downside.
In the end, the conclusion seems to be that in a regular hydroponics system where pH increases generally happen towards the upside it is better to use carbonate as a buffering agent than to use citrate or phosphate although phosphate at its regular concentration in hydroponic does provide some buffering against pH moves (without phosphate increases are much more dramatic). For this reason I believe that a phosphate/carbonate buffer seems to be the best choice for most hydroponic growers, taking care to keep the concentrations at levels that do not cause precipitation or phytotoxicity problems.