HydroBuddy v1.9, MacOS binary, new EC model, many bug fixes and more!
Today I am releasing a new version of HydroBuddy (v1.9) which contains many suggested and needed improvements from the previous version of the software. In this post I want to discuss the changes within this release and how they will affect the way things are done in the program. Some big changes have been implemented so make sure you go through the list below if you want to use this new version. Thanks to all of you who contributed your suggestions about HydroBuddy and/or reported bugs to me.
Here is the list of changes in this version:
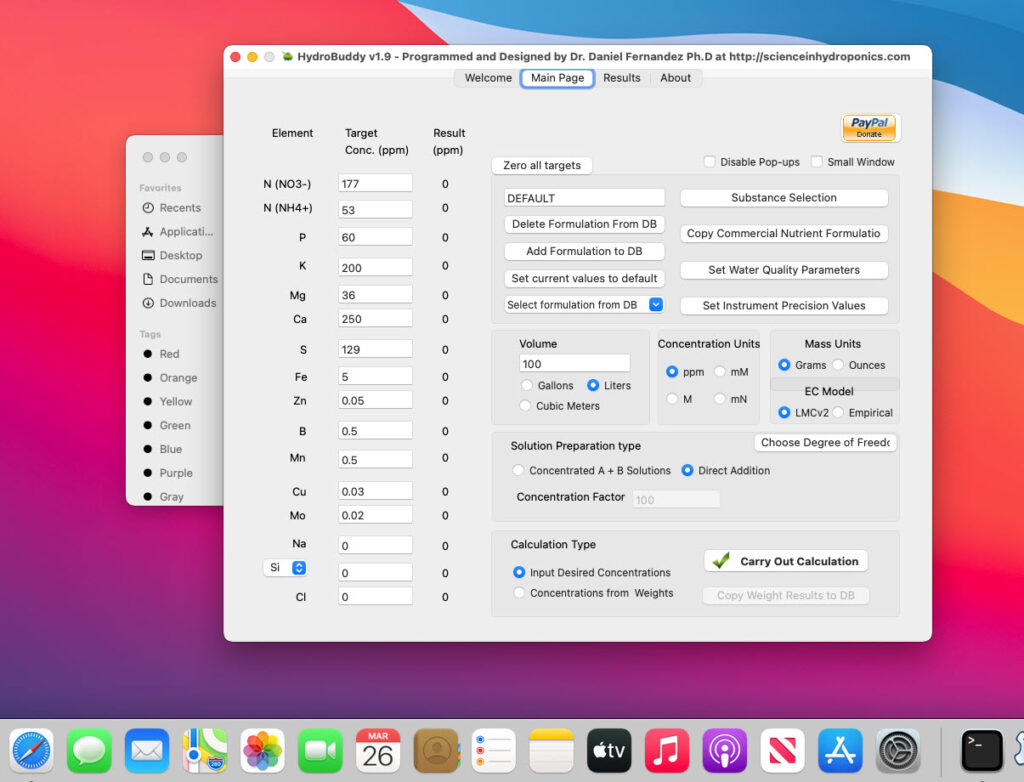
- A MacOS binary compiled in Big Sur 11.0.1 has been released.
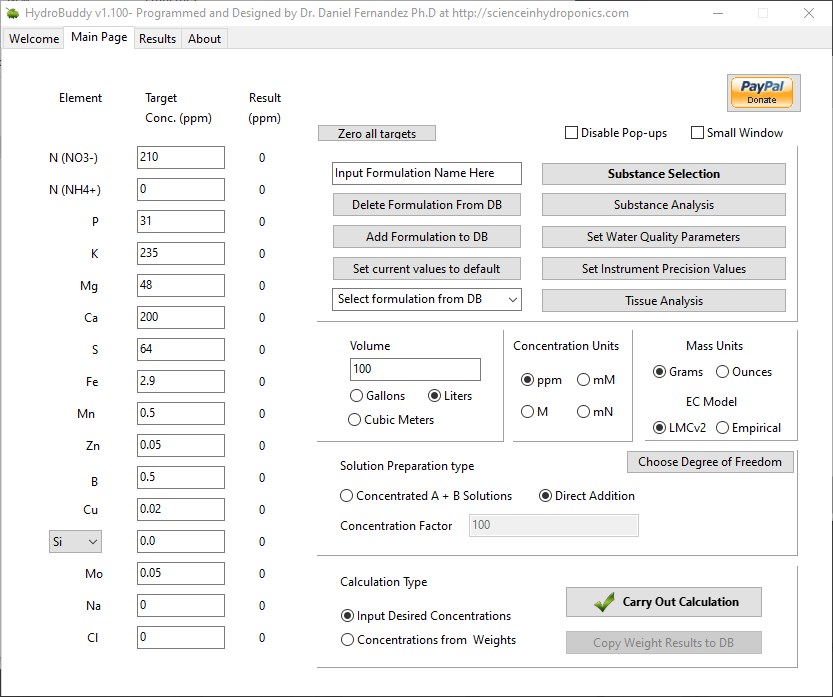
- Ability to make any formulation the “default” formulation. This selected formulation is loaded when the software is started.
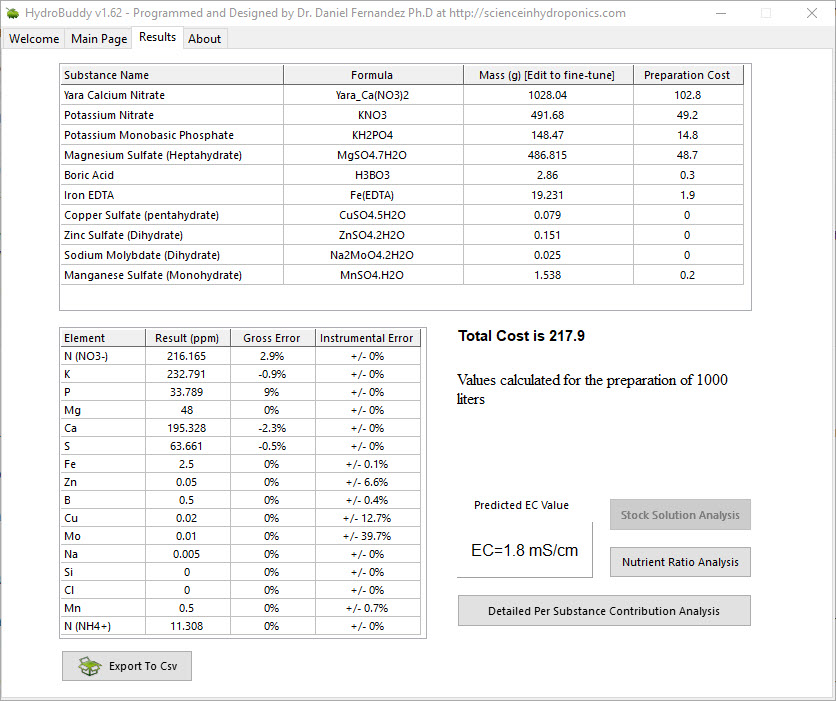
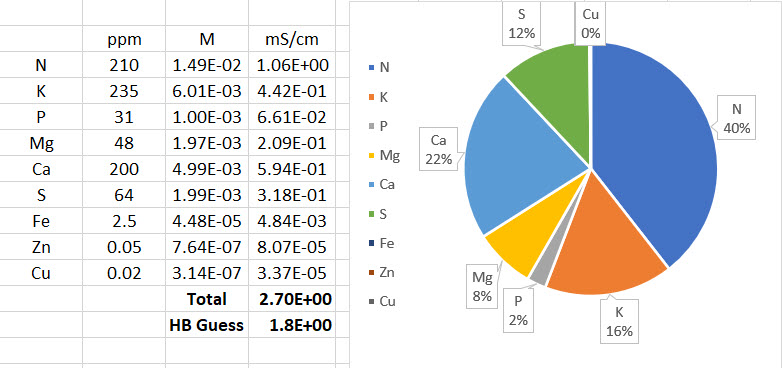
- The LMC conductivity model has now been replaced with LMCv2 which is an important improvement. See here to learn more. The LMCv2 model now adjusts conductivity based on each specific ion’s charge and the overall ionic strength of the solution. It now includes no arbitrary terms.
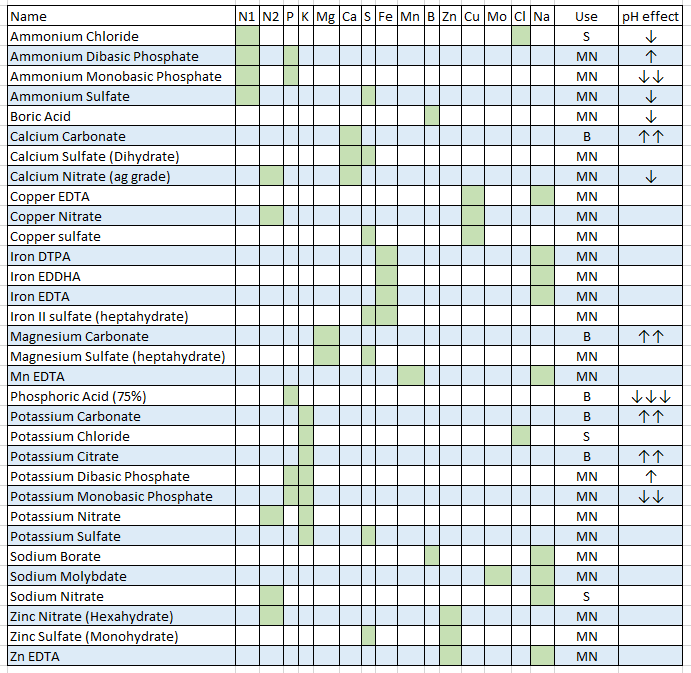
- The treatment of liquids/solids in the program has now been changed. Instead of specifying liquid or solid (and the program having to make assumptions) users can now select whether the percentages and substance amounts are going to be either in g and w/w% or in mL and w/v%. This should simplify the interpretation of results and the addition of substances.
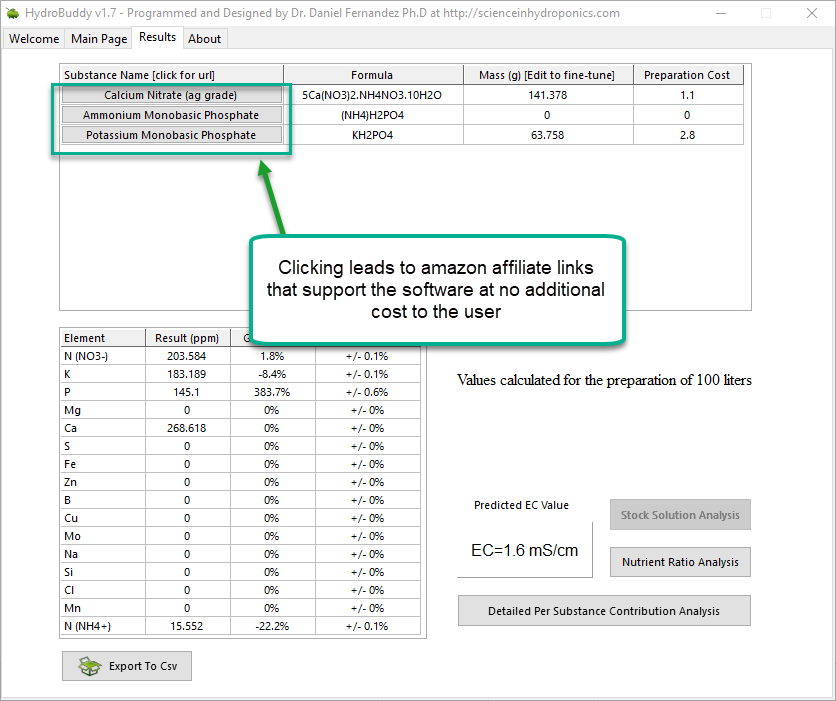
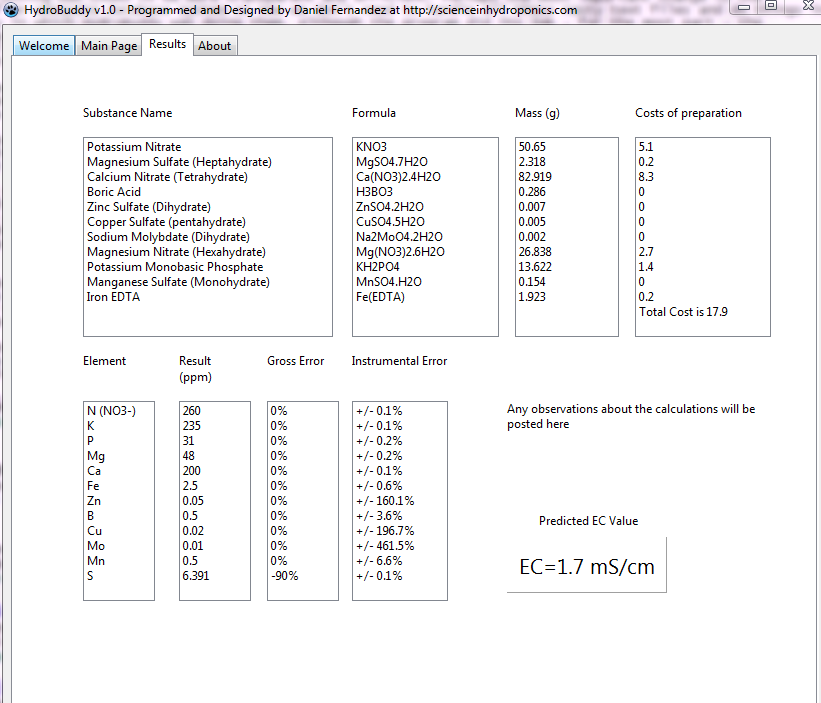
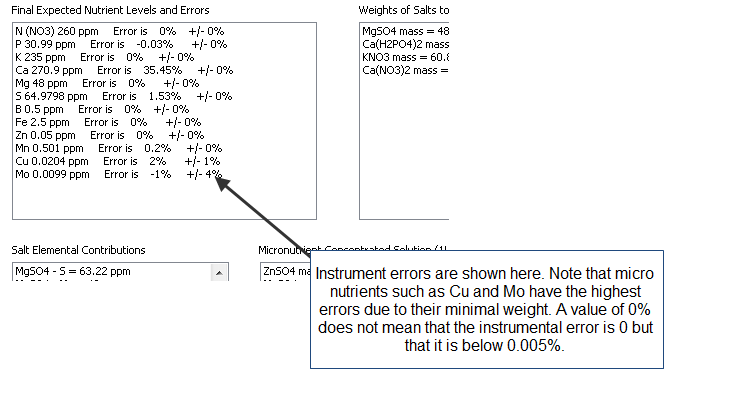
- An additional column has now been added in the results page to specify the unit of the amount being calculated. When a user wants a substance’s contribution to be calculated in mL, the appropriate unit will be shown here.
- When adding a new substance, all fields are reset to null values (previously the program kept the values from previously opened/updated substances).
- Density has now been eliminated as a variable used in the program since it is not needed if there is no cross between w/w% and w/v% calculations. It is only kept in the “Copy commercial nutrient formulation” dialogue.
- An error where P and K were mixed up in the product comparison window of the “Copy commercial nutrient formulation” function has now been fixed.
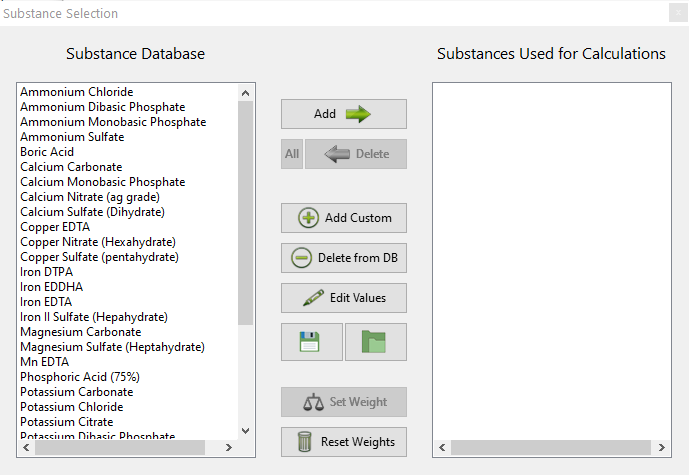
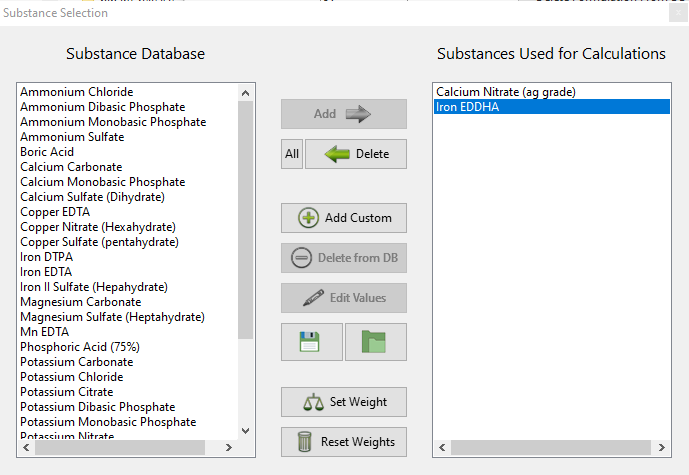
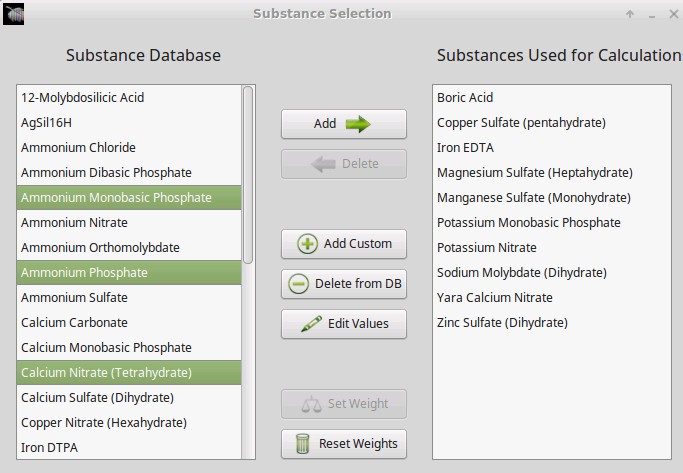
- The wording of options in the “Substance selection” dialogue has been changed so that the buttons better describe what they do. For example the “Delete” button has now been changed to “Do not use”.
- Two buttons have been added next to the EC model prediction in order to allow users to increase or decrease the EC by adjusting all nutrient concentrations by +5%/-5%. This will allow you to see how nutrient concentration changes affect conductivity in a straightforward manner.
The above modifications are now committed to the github repository as well. Feel free to take a look if you’re interested in how any of the above variations were coded into the program.